🌡 nm to Kelvin Converter
Convert nanometer wavelength to Kelvin temperature using Wien's Displacement Law
| Source / Application | Wavelength (nm) | Temperature (K) | Temp (°C) | Spectrum Region |
|---|---|---|---|---|
| UV Germicidal Lamp | 254 | 11,409 | 11,136 | UV-C |
| UV-A Blacklight | 365 | 7,939 | 7,666 | UV-A |
| Violet Light | 400 | 7,244 | 6,971 | Visible |
| Blue Light | 450 | 6,439 | 6,166 | Visible |
| Cyan Light | 490 | 5,914 | 5,641 | Visible |
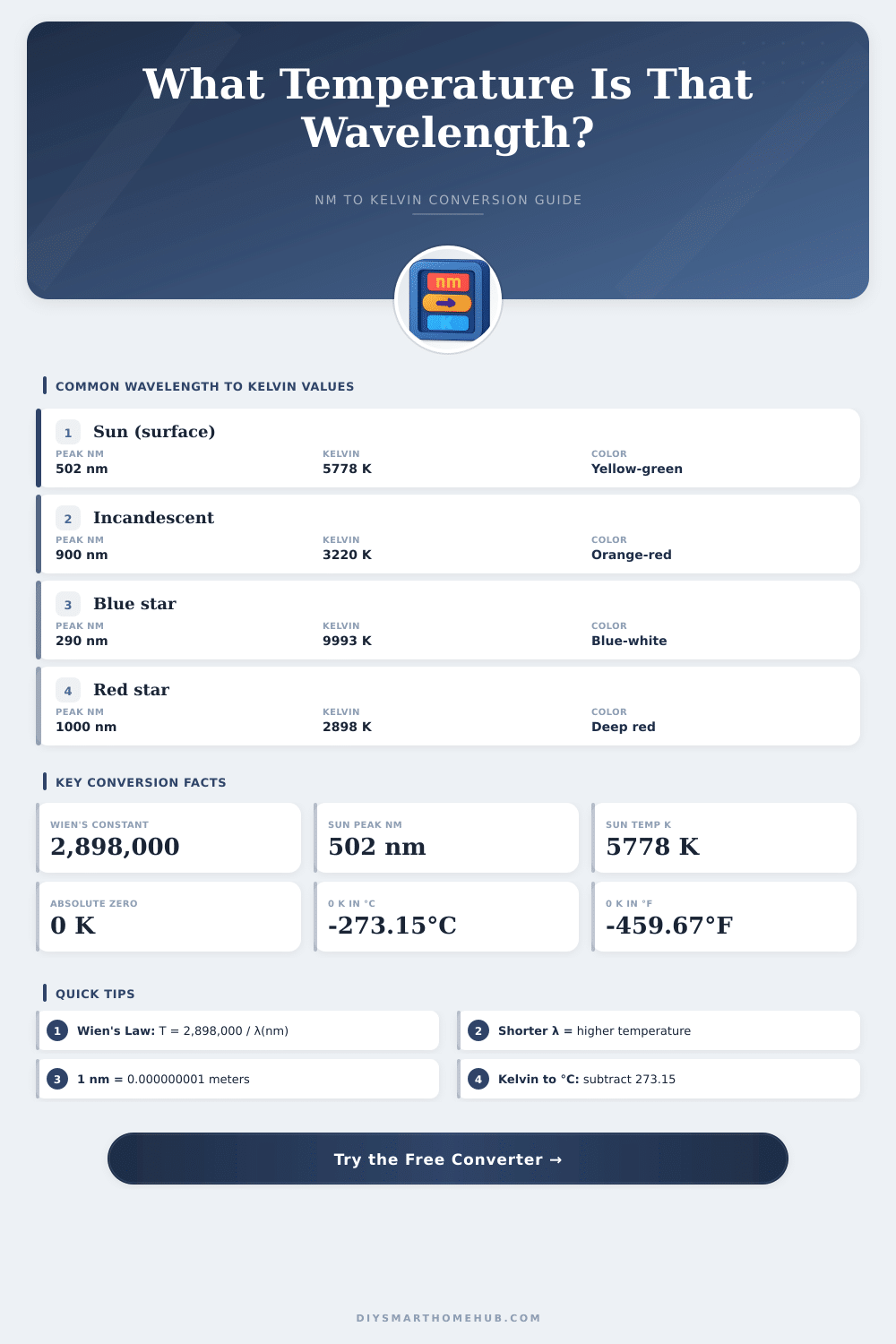
| Sun Peak / Yellow-Green | 502 | 5,773 | 5,500 | Visible |
| Green Laser | 532 | 5,448 | 5,175 | Visible |
| Yellow Sodium | 589 | 4,920 | 4,647 | Visible |
| Red LED | 660 | 4,391 | 4,118 | Visible |
| Deep Red / Far Visible | 700 | 4,140 | 3,867 | Visible |
| IR Night Vision | 850 | 3,409 | 3,136 | Near-IR |
| IR Remote Control | 940 | 3,083 | 2,810 | Near-IR |
| Nd:YAG Laser | 1,064 | 2,724 | 2,451 | Near-IR |
| Incandescent Peak | 900 | 3,220 | 2,947 | Near-IR |
| CO2 Laser | 10,600 | 273.4 | 0.3 | Mid-IR |
| Unit | Symbol | In Meters | Equivalent in nm |
|---|---|---|---|
| Nanometer | nm | 1 × 10⁻⁹ m | 1 nm |
| Angstrom | Å | 1 × 10⁻¹⁰ m | 0.1 nm |
| Micrometer | μm | 1 × 10⁻⁶ m | 1,000 nm |
| Millimeter | mm | 1 × 10⁻⁹ m (wait: 10⁻⁹ is nm) 1 × 10⁻³ m | 1,000,000 nm |
| Centimeter | cm | 1 × 10⁻₂ m | 10,000,000 nm |
| Meter | m | 1 m | 1,000,000,000 nm |
| Kelvin (K) | Celsius (°C) | Fahrenheit (°F) | Notes |
|---|---|---|---|
| 0 K | −273.15 °C | −459.67 °F | Absolute Zero |
| 273.15 K | 0 °C | 32 °F | Water Freezing Point |
| 373.15 K | 100 °C | 212 °F | Water Boiling Point |
| 2,898 K | 2,625 °C | 4,757 °F | 1000 nm peak (Wien) |
| 3,220 K | 2,947 °C | 5,336 °F | Incandescent bulb (~900 nm) |
| 5,778 K | 5,505 °C | 9,941 °F | Sun surface (502 nm peak) |
| 10,000 K | 9,727 °C | 17,540 °F | Blue-white star |
| 29,000 K | 28,727 °C | 51,740 °F | Hot blue star (~100 nm) |
| Star Class | Color | Peak Wavelength (nm) | Surface Temp (K) |
|---|---|---|---|
| O-type | Blue-white | < 300 nm | > 30,000 K |
| B-type | Blue-white | 290 – 390 nm | 10,000 – 30,000 K |
| A-type | White | 390 – 490 nm | 7,500 – 10,000 K |
| F-type | Yellow-white | 490 – 560 nm | 6,000 – 7,500 K |
| G-type (Sun) | Yellow | 502 nm | 5,200 – 6,000 K |
| K-type | Orange | 600 – 800 nm | 3,700 – 5,200 K |
| M-type | Red | > 800 nm | < 3,700 K |
In first look, convert between nanometroj and Kelvin could seem as if you try to mix oil with water. But really? It is much more easy when one gets the main idea.
The main point is, that those two units describe entirely different causes. Nanometroj tell you about a particular wavelength of light, so, about one certain colour. Kelvin otherwise estimates the warmth, that black body gives off.
Can you convert nanometres to Kelvin?
That warmth directly determines, that light spectrum the object generates.
Here where it becomes interesting. There do not exist real one-for-one conversion between nanometroj and Kelvin. Those two units simply do not work like this.
Kelvin estimates the whole colour range of a light source, not only one wavelength. Here this said, it however helps to estimate, where the peak of wavelengths sits in that spectrum. Picture it thus: the Kelvin-temperature describes the light colours, that black body gives off…
Think about warm metal, that shines only because of warmth, without any chemical colouring.
Even so exists mathematical tie between them. It calls Wien-key, and here this it means: one takes 2 897 768 and shares it by the colour temperature in Kelvin to receive nanometroj. Enter the colour temperature of a light source and count, you find the most intense wavelength in that spectrum.
It is not perfect direct conversion, but it gives good notion about the probable peak of wavelengths in a light source.
Calculator removes the troubles from all that. Enter the Kelvin-temperature, receive the wavelength in nanometroj, and the device gives the value, that you need. Lot simpler then compile the numbers self every time.
Many folks try to create simple table, that binds colour temperature with nanometroj. But do that purely and usable? Surprisingly hard.
It is like to mix random colours in tin and later decide, is the mix red, orange, yellow, green, blue, indigo or violet. The answer simply is not easy. Folks bother to find chart, that shows Kelvin and nanometroj side by side, although the theme appears quite commonly.
One more cause to recall. Kelvin also estimates molecule movements in entirely other context. In zero Kelvin, molecules stop moving entirely.
That is separate usage from light and colour work, that only shows, how big a range of uses the Kelvin-unit has.
You also will find online tools for convert Newton-metres to Kelvin or Newton-grade to Kelvin. Fast search shows, that one Newton-grade matches around 3,03 Kelvin. That comes up sometimes, when one hunts converters, although it differs from the nanometro-sight.
Because of that, for anyone, that really works with light and colours, the tiebetween nanometroj and Kelvin stays the most practical resource.