🌡️ Calories to Kelvin Converter
Convert thermal energy (calories & kilocalories) to equivalent Kelvin temperatures using Boltzmann & thermodynamic formulas
| Energy Input | Joules (J) | Kelvin (K) – Mono | °Celsius (°C) |
|---|---|---|---|
| 0.001 cal | 0.004184 J | 2.02 × 10⁻²⁰ K | −273.15 °C |
| 0.01 cal | 0.04184 J | 2.02 × 10⁻¹⁹ K | −273.15 °C |
| 0.1 cal | 0.4184 J | 2.02 × 10⁻¹⁸ K | −273.15 °C |
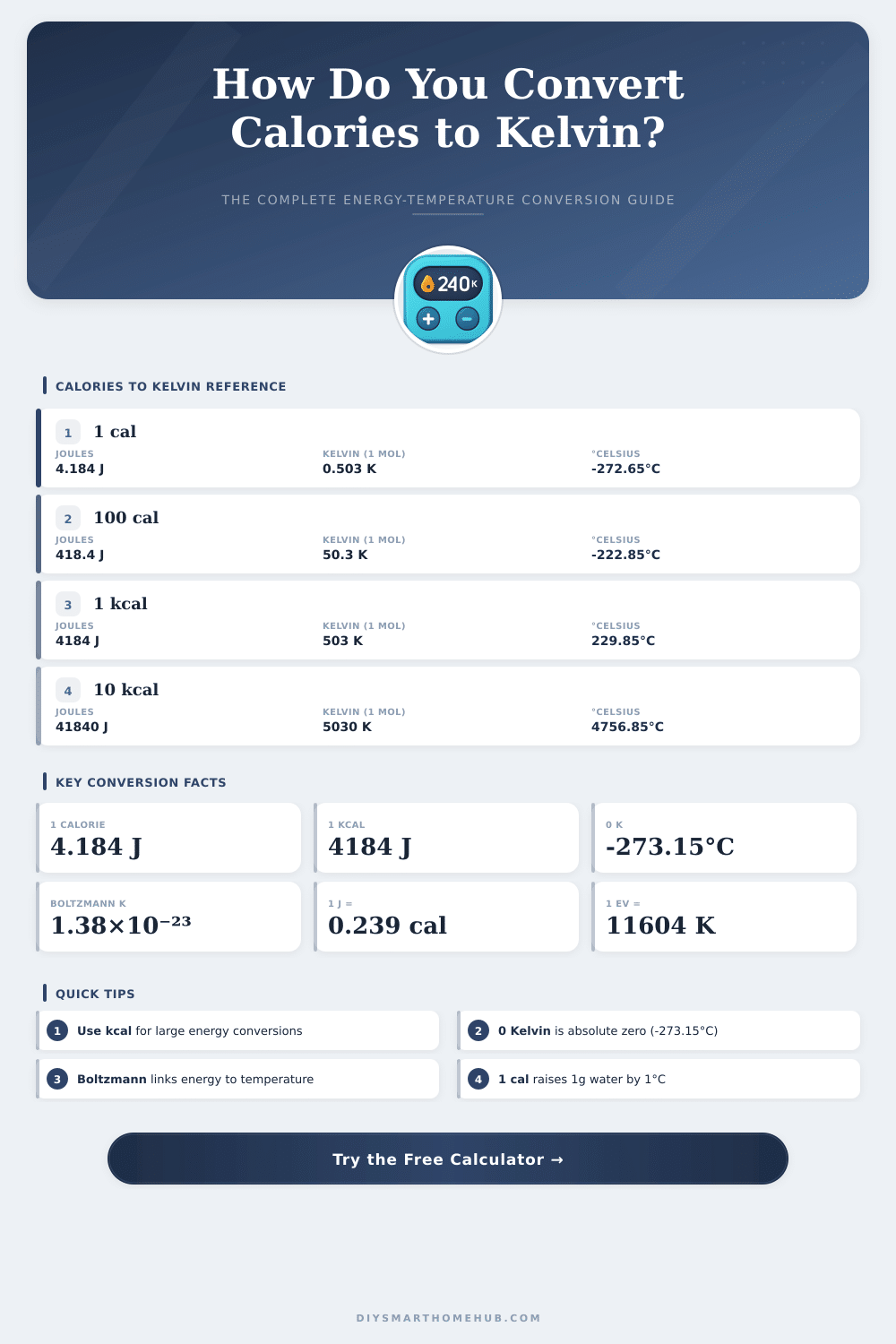
| 1 cal | 4.184 J | 2.02 × 10⁻¹⁷ K | −273.15 °C |
| 10 cal | 41.84 J | 2.02 × 10⁻¹⁶ K | −273.15 °C |
| 100 cal | 418.4 J | 2.02 × 10⁻¹⁵ K | −273.15 °C |
| 1 kcal | 4,184 J | 2.02 × 10⁻¹⁴ K | −273.15 °C |
| 10 kcal | 41,840 J | 2.02 × 10⁻¹³ K | −273.15 °C |
| 100 kcal | 418,400 J | 2.02 × 10⁻¹² K | −273.15 °C |
| 2000 kcal | 8,368,000 J | 4.04 × 10⁻¹¹ K | −273.15 °C |
| Method | Formula | Result (K) | Best Used For |
|---|---|---|---|
| Monatomic Gas (1 particle) | T = 2E/(3Nk) | 2.02 × 10²³ K | Single-atom KE |
| Diatomic Gas (1 particle) | T = 2E/(5Nk) | 1.21 × 10²³ K | Diatomic molecules |
| Boltzmann (E = kT) | T = E/(Nk) | 3.03 × 10²³ K | Characteristic temp |
| Monatomic (1 mole) | T = 2E/(3nR) | 335.2 K (62°C) | Molar thermodynamics |
| Diatomic (1 mole) | T = 2E/(5nR) | 201.1 K (-72°C) | Molar diatomic gas |
| Boltzmann (1 mole) | T = E/(nR) | 502.8 K (230°C) | Molar characteristic |
| From Unit | Joules (J) | Calories (cal) | BTU |
|---|---|---|---|
| 1 Calorie (cal) | 4.184 J | 1 cal | 0.003969 BTU |
| 1 Kilocalorie (kcal) | 4,184 J | 1,000 cal | 3.9683 BTU |
| 1 Joule (J) | 1 J | 0.23901 cal | 0.000948 BTU |
| 1 Kilojoule (kJ) | 1,000 J | 239.01 cal | 0.94782 BTU |
| 1 BTU | 1055.06 J | 252.16 cal | 1 BTU |
| 1 Electron Volt (eV) | 1.602 × 10⁻¹⁹ J | 3.829 × 10⁻²° cal | 1.518 × 10⁻²² BTU |
| Reference Point | Kelvin (K) | Celsius (°C) | Fahrenheit (°F) |
|---|---|---|---|
| Absolute Zero | 0 K | −273.15 °C | −459.67 °F |
| Water Freezes | 273.15 K | 0 °C | 32 °F |
| Room Temperature | 293.15 K | 20 °C | 68 °F |
| Body Temperature | 310.15 K | 37 °C | 98.6 °F |
| Water Boils | 373.15 K | 100 °C | 212 °F |
| Solar Surface | 5,778 K | 5,505 °C | 9,941 °F |
Use Monatomic Ideal Gas for noble gases (He, Ne, Ar). Use Diatomic for air molecules (N₂, O₂). Use Boltzmann (E=kT) to find the characteristic temperature where thermal energy equals your input energy. Use Molar settings for real-world chemistry problems involving measurable amounts of substance.
When converting macroscopic calorie amounts (like a food calorie) for a single particle, the resulting Kelvin is enormous — that's physically correct but not physically realistic (no object reaches 10²³ K). Switch to 1 Mole of particles for thermodynamically meaningful temperature results that match real-world gas behavior equations.
Convert between calories and kelvinoj maybe seem strange at first. That pairs of units measure entirely different things even so exist resources and equations that help with the change. Many websites offer tools for range different units, also of calories to kelvinoj, by means of many factors.
Similarly works the course of kelvinoj to calories about nutrition.
How to convert calories and kelvin
The equation for changing kelvinojn in nutritious calories truly surprise. One kelvino match around 3,298 × 10 in the power minus 27 nutritious calories. So kelvino are around 3,033 × 10 in the power 26 times more small than nutritious calorie.
Such numbers are surprisingly small or huge, what only stresses the big difference between those units according to size.
Exist conversions for the IT-calorie to kelvino and for the IT-kilokalorio to kelvino. They take in thought various meanings of the calorie in sciences. Also, entropy one sometimes measures by means of calories for kelvino, and the molara varmkapablo can appear as calories for mol kelvino.
One commonly meets them in physics and in math.
Talking about close conversions, one calorie for kelvino value around 0,00418 kilojuloj for kelvino. Mistakes during rounding can grow, so always value checking the results. When deal about bigger values, the change of kilojuloj for kelvino to calories for kelvino result for example in 9 kilojuloj for kelvino matching 2151 calories for kelvino, and 10 kilojuloj four kelvino matching 2390 calories for kelvino.
Particular varmkapablo form other fields, where calories and kelvinoj meet. For it one uses calculators with units as calorie for gram kelvino, kilokalorio for kilo kelvino and thermal calorie for gram kelvino. Beside them one uses also ĵul-based and watt-based units bound to kelvinoj.
Big reason to know is the difference between calories and kilokalorioj. It deals about the capital letters. The big “Cal” appear on nutritious labels in United States, while “kcal” one uses in Europe.
Also, the small k means kilo, what wants to say 1000, to the big K stand for kelvino, named after Lord Kelvin. If one mixes them, easily comes confusion.
In chemistry calories one still uses a lot. The energy, that reaction releases in water solution and express by means of kilokalorioj for mol, almost match to the focus in mol because of liter, multiplied by means of the temperature change in kelvinoj or grades Celsio. The theme for gases, 8,314 joules for kelvino, one can say also in calories for kelvino.
One counts energy by means of the relation, where it matches half-triple of the molnombro, times the gaseous theme, times the temperature in kelvinoj.
The units calorie for kelvino matter for understanding energyefficiency in science about foods, in environmental studies and in engineering.